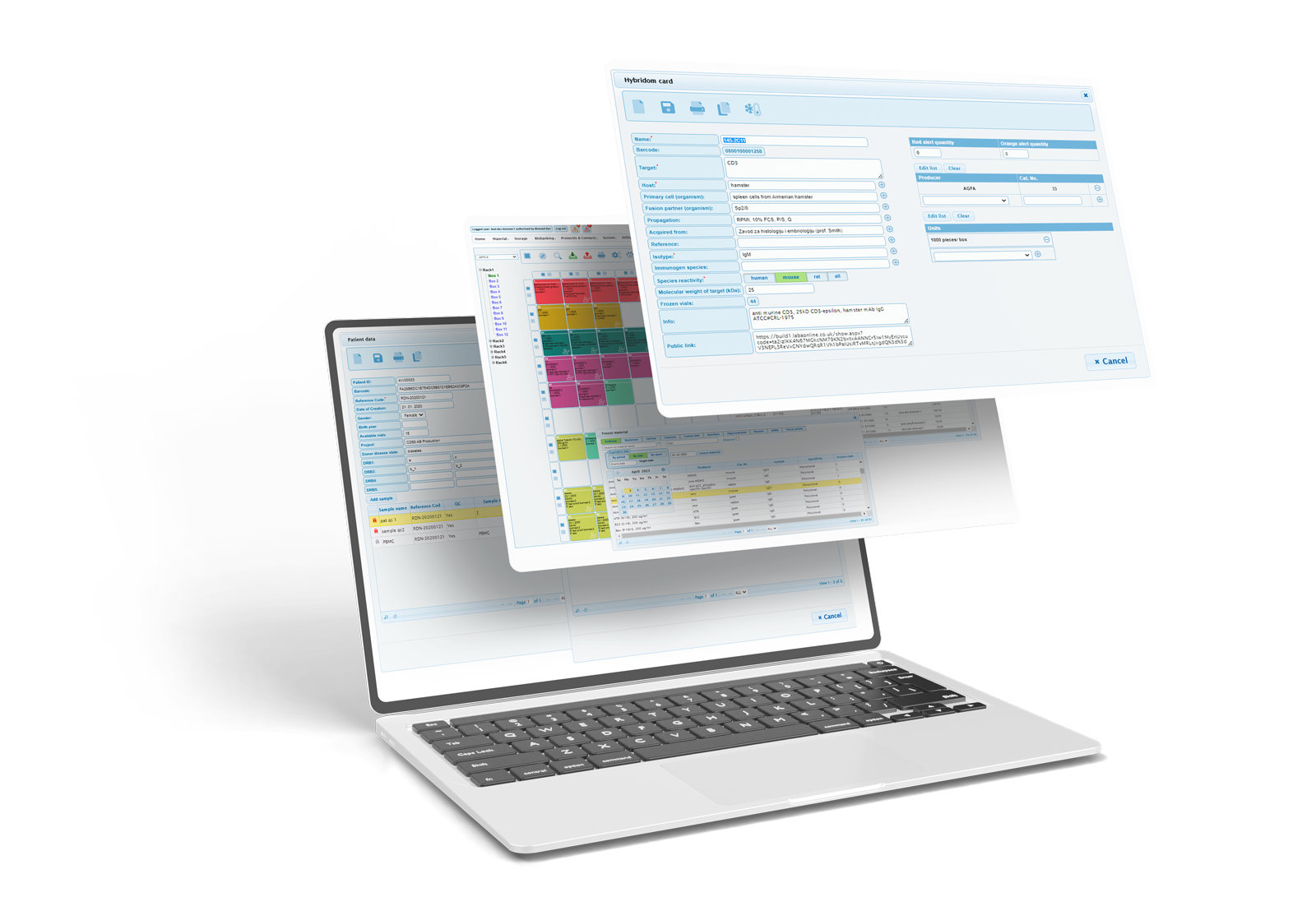
Biobanking Lims System
LABORATORY SOFTWARE
You can use our Biobanking LIMS:
- To manage reagents
- To create a patient repository
- To find vials in freezers
- To store your experiments
Want a custom LIMS? Just tell us what you want, and we’ll develop the features you need.


About Biobanking
LIMS Laba
LABA is a laboratory and biobanking management system that allows users to describe, locate and use materials, patient samples and storage facilities. It is an integrated system accessed by one login.
Who can use our Biobanking LIMS software:
- Biomedical Laboratories
- Hospitals
- Forensic Laboratories
- Diagnostic Laboratories
Request a 14-days free trial or even better: schedule a live demo to see LABA in action.
- One login to manage reagents, biobanking, storage facilities and knowledge base.
- A relational database and innovative functionalities provide non-redundant, evaluated and fast data entry.
- Intuitive interfaces with well-structured forms and easy recognisable functions.
- Each team member’s access and rights are finely tuned according to current lab needs.
- Track stored material and protocols by username.
- All the lab’s knowledge is accumulated ensuring fast research progress.
- Cloud option for labs requiring the latest LABA version.
- Self-hosting option for labs requiring software customisation.
- Quantity and expiry alarm can be assigned to any material.
- Expiry alarm is defined per date and automatically assigned to orange or red category.
- Orange and red quantity alarms are user-defined for any chosen material.
What is a
LIMS system?
A Laboratory Information Management System (LIMS) helps improve lab efficiency and productivity. It helps lab scientists track data associated with their experiments, instruments, samples and laboratory workflows.
The use case and laboratory could have specialised features for research and development (R&D) laboratories, bioanalytical laboratories, process development, and manufacturing labs.
How do LIMS systems work?
LIMS systems allow technicians and scientists to enter detailed information about the samples they are working with. Examples of this information include:
Location of test conducted
Batch material
Date and time
Inspection number
Other important details
Today, many businesses have to store large amounts of data for effective operations, which is most apparent in a laboratory. With the advancement in technology, there has been an increase in the amount of data we hold in labs and the technical details.
The complexity of data means that spreadsheets aren’t enough to store laboratory records as they should, which is where a LIMS system comes into play.
A sample may undergo several processes throughout tests and collect several data along the way. LIMS software provides an easy way to organise and sort this information. All of the information in LIMS is safe, secure, and easily accessible.
LIMS would typically generate a barcode after you enter the initial sample data so that other staff members can quickly scan and add further information as they get it.
Every time a user scans the same code, they get access to updated data that they can use to continue their testing processes. Apart from ensuring data safety, you can also use LIMS to track tests, plan updates and transfer test results to other systems in a chain.
LIMS eliminates the need to manually record data, reducing human error as the system will automatically capture data.
What Is the difference between a LIMS and database?
From the definition of LIMS, you might wrongly assume that it is a database. A LIMS is usually a standalone product that you can integrate with databases to quickly and accurately validate samples and then send out the results as needed. Also, LIMS could have an interface where you can request analysis and get results for other customers of laboratory services.
Benefits of a LIMS systems
LIMS are constantly updating their features to meet the changing needs of labs. Already, LIMS has several benefits, including:
Apart from samples, you can also use LIMS to track all the consumables and reagents that enter a lab. You can use it to record the expiration dates and prevent the use of expired goods, keep track of recipes including the reagents, get records of who made them, track volumes as you use the reagents and notify users when it is time to reorder.
This means the LIMS knows your inventory, and it can therefore reduce over-ordering, which could lead to waste and increased costs. The LIMS will help you eliminate unplanned reagent depletion, which may cause project overruns and other work delays.
The most significant benefit by far is tracing a reagent or consumable when the need arises. If there is any challenge with a particular reagent, you can use the LIMS to identify which samples are affected and retest them appropriately.
The most basic Laboratory Information Management systems allow you to log in samples, record their exact location down to the site, lab, freezer, shelf, box and cell. You will also record and view the sample’s journey in each step of the process through the lab workflow you are performing.
You would typically divide samples into aliquots, and LIMS will track the model and the result obtained at each aliquot. It also gives you a consolidated view at the patient, sample, project or batch level as your workflow demands.
LIMS is one of the most excellent tools for aggregating data. It carefully tracks all the changes throughout the sample’s lifecycle and gives a comprehensive audit trail of every action you performed on each model.
Many laboratories have to comply with many regulations and practices at different levels. Some of these regulations are worldwide, while others are localised. Some laboratory practices include GxP and ISO 17025 that cover how you handle data, like the 21 CFR Part 11 and all the data integrity guidelines.
In many regions, compliance necessitates the use of LIMS for their operations. LIMS helps keep laboratories compliant with the practices that reduce human error and ensure you handle samples with the utmost care.
Apart from samples, you can also use LIMS to track all the consumables and reagents that enter a lab. You can use it to record the expiration dates and prevent the use of expired goods, keep track of recipes including the reagents, get records of who made them, track volumes as you use the reagents and notify users when it is time to reorder.
This means the LIMS knows your inventory, and it can therefore reduce over-ordering, which could lead to waste and increased costs. The LIMS will help you eliminate unplanned reagent depletion, which may cause project overruns and other work delays.
The most significant benefit by far is tracing a reagent or consumable when the need arises. If there is any challenge with a particular reagent, you can use the LIMS to identify which samples are affected and retest them appropriately.

Should you have any questions, don’t hesitate to contact us.
We shall reply in 24 hours.
Please check the spam folder.





